Amtec Guide to Cathodic Protection in Ballast Tanks and Cargo Holds
Cathodic Protection has been employed in metal ships since Sir Humphrey Davy first used the method to protect several British Naval vessels in 1824.
As explained earlier, the dissolution of steel is an anodic process which takes place when metallic iron loses two electrons and becomes an iron ion in solution. If the potential of a corroding object is lowered (i.e. it is made more electronegative) to the reversible potential of the anodic reaction, then the metal dissolution will stop, since the rate of dissolution will exactly balance the rate of re-deposition.
When measured against a reference electrode such as a Saturated Calomel Electrode (S.C.E.), the reversible potential of corroding steel referred to above, is in the order of -860mV (S.C.E.). The measured potential for mild steel in sea water (which is a combination of the potential of the anodic and cathodic reactions taking place) is in the order of –640mV (S.C.E.) measured with the same reference electrode.
To protect steel successfully using cathodic protection, it is therefore only necessary to lower its potential by around a quarter of one volt (250mV).
Cathodic protection using sacrificial anodes produces a decrease in the potential of the ship by connecting the vessel to a metal which takes up a reversible potential of less than –850mV (S.C.E.) and allowing the sacrificial metal to produce the electrons rather than the corrosion reaction of the steel.
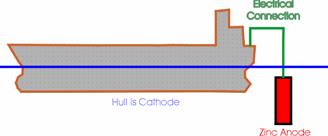
Figure 6-1 Sacrificial Cathodic Protection schematic.
Common choices for the sacrificial anodes are zinc, aluminium and their alloys. Both zinc and aluminium produce potentials of less than –1000mV (S.C.E.). Aluminium has to be alloyed with other metals such as tin in order to make it corrode freely in service.
Magnesium anodes cannot be used in ballast tanks because they generate hydrogen in use and can have deleterious effects on some ballast tank coatings. Aluminium anodes have limited use in tankers as they can produce a spark hazard if they fall or are dropped from significant heights.
The protection current available from the zinc and aluminium anodes are similar. Zinc requires 11.3Kg (25 pounds) of material to produce one Ampere-year of current, whilst 8.2Kg (18 pounds) of aluminium is necessary.
In addition to protection via an electrochemical potential change, sacrificial anodes also protect by forcing the cathodic reaction to occur on the steel. The cathodic reaction reduces oxygen and water to generate alkaline hydroxyl ions as described previously. The production of calcareous deposits initially enhances the protection effect.
Sacrificial systems in heavy weather holds and ballast tanks are not capable of working during the period when the tanks are empty. This may constitute 60% of the vessel usage pattern. Unfortunately, steel corrodes most rapidly when it is damp with a conducting film over the surface rather than fully immersed and the sacrificial anode system cannot act directly at this time. The CP system does have an indirect effect during this period before the tank dries, as the retained hydroxyl ions work to keep the steel surface passive and the calcareous deposits act as a barrier to oxygen.
In very general terms, a steel surface which is coated with a good quality paint will drain in the order of 5mA/m2 from a cathodic protection system and an exposed steel surface will require 110mA/m2 for protection. Consequently, the current requirement for a tank will increase with time as the extent of coating breakdown (and thus exposed metal area) increases.
The efficiency of a cathodic protection system can be assessed by examination of both the condition of the sacrificial anodes and the nature of the calcareous deposit produced. Photograph 6-1 below shows an anode functioning normally. The surface is covered with a compact layer of coherent white deposit.

Photograph 6-1. Normally working anode.
Photograph 6-2 below shows an overworked zinc anode. The deposit is very loose and is easily removed from the surface of the anode.

Photograph 6-2. Overworked zinc anode.
Photograph 6-3 below shows an overworked aluminium anode. The gelatinous deposits dripping from the anode are indicative of excessive current drain.

Photograph 6-3. Overworked aluminium anode.
Photograph 6-4 below shows a ballast tank where the coating barrier properties are failing and calcareous deposits are forming throughout the tank.

Photograph 6-4. Heavy calcareous deposits.
The siting of anodes is also important as they must be placed clear of any overhanging stiffeners whenever possible, to aid the ionic flow around the anodes and prevent incorrect current flow. Wrongly sited anodes can result in severe blistering of the coating in their immediate vicinity and figure 6-6 below is an example of this effect.

Photograph 6-5. Poorly sited anode.
On exterior hulls, it is also possible to use an impressed current system in the same way that coupling mild steel to zinc results in a flow of electrons to the mild steel to prevent metal loss. In this system, an auxiliary anode made from a non-consumable material, platinised niobium, replaces the zinc or aluminium anode of the sacrificial system. Areas immediately around the impressed current anodes can suffer from blistering and a thick layer of anode shield material is applied to prevent the loss of the paint.

Figure 6-2. Schematic diagram of an impressed current system.
The electrodes in such an impressed current system are capable of drawing very high currents locally and thus producing copious quantities of oxygen, hydrogen and chlorine. This is totally unacceptable in holds and tanks and consequently impressed current systems are not used in these areas.


